Stem cell therapy for cornea repair represents a groundbreaking advancement in the field of ocular medicine, offering new hope for patients suffering from corneal injuries previously deemed untreatable. At the forefront of this innovation is the cultivated autologous limbal epithelial cell (CALEC) procedure, which utilizes a patient’s own stem cells to restore the corneal surface effectively. Recent clinical trials have shown an impressive success rate of over 90% in visually restoring the cornea, presenting a safe alternative for corneal injury treatment. By focusing on renewing the eye’s natural defenses, this therapy addresses the limbal epithelial cell deficiency that results from various types of eye damage. As research continues to expand on stem cell clinical trials, the promise of improved outcomes for vision rehabilitation is becoming more real for countless individuals in need.
Innovative solutions for eye damage repair are emerging through advancements in regenerative medicine, particularly with stem cell strategies designed for corneal restoration. The groundbreaking CALEC method exemplifies how modern techniques, such as limbal epithelial cell transplantation, can convert a patient’s own healthy eye cells into a viable graft for injured eyes. This promising approach to corneal injury treatment not only revitalizes hope among patients but also initiates a new era in ocular therapies. By harnessing the body’s natural healing capabilities, researchers are paving the way for comprehensive treatment options that could finally address long-standing visual impairments. As clinical studies advance, they aim to enhance the standard practices in ophthalmology and broaden access to these pivotal therapies.
The CALEC Procedure: A Breakthrough in Corneal Repair
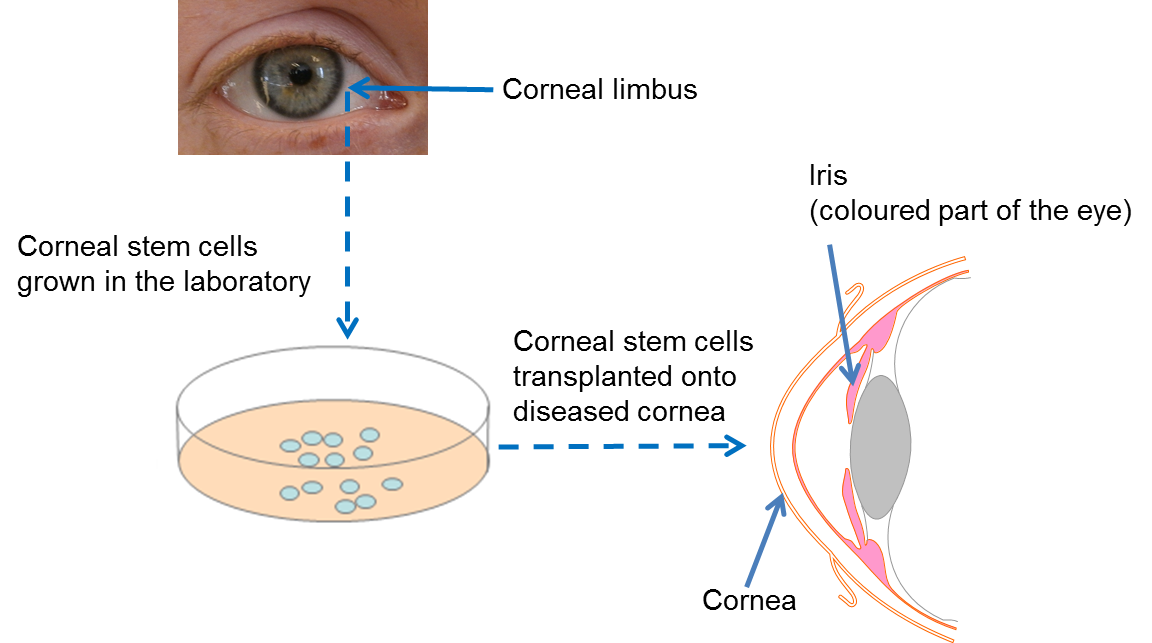
The cultivated autologous limbal epithelial cells (CALEC) procedure marks a significant advancement in corneal repair, especially for patients with severe corneal injuries. By employing stem cells harvested from a healthy eye, clinicians are now able to reconstruct the limbal epithelial cells that are vital for the cornea’s integrity. This process involves a meticulous biopsy to extract these stem cells, which are then cultivated in a lab to produce a tissue graft ready for transplantation. The pioneering efforts by Ula Jurkunas and her team at Mass Eye and Ear have not only demonstrated the procedure’s safety but have produced promising results, with rates of corneal surface restoration reaching 90% in clinical trials.
The CALEC procedure serves as a beacon of hope for those suffering from corneal injuries that were previously deemed untreatable. Traditional options like corneal transplants often come with limitations, particularly for individuals who have exhausted their supply of healthy limbal stem cells due to injury or illness. Through the innovative approach of cultivating stem cells from the patient’s unaffected eye, CALEC offers personalized treatment that addresses this stark clinical need. As interest in this revolutionary therapy grows, further studies could enhance its applicability, potentially paving the way for broader inclusion criteria and even allogeneic applications to assist patients with bilateral corneal damage.
Understanding Corneal Injury and Limbal Stem Cell Deficiency
Corneal injury can manifest from various sources, including chemical burns, infections, or trauma, leading to a debilitating condition known as limbal stem cell deficiency. This condition prevents the natural regeneration of the cornea’s protective epithelial layer, leaving patients vulnerable to severe pain and visual impairment. In many cases, the damage is so profound that conventional corneal transplantation fails to provide a viable solution. Thus, a comprehensive understanding of the underlying mechanisms of corneal injury is critical for developing effective treatments and optimizing outcomes for affected individuals.
The absence of healthy limbal epithelial cells severely hinders the eye’s ability to heal, making it imperative for researchers to explore innovative therapeutic options such as limbal epithelial cell transplantation. The introduction of CALEC into the clinical landscape not only redefines standard treatment protocols but also emphasizes the importance of preserving healthy eye tissue for potential regenerative therapies. The dynamic interplay between corneal injuries and limbal stem cell health underlines why targeting these cells is crucial in developing effective corneal injury treatments.
Potential of Stem Cell Therapy in Eye Damage Repair
Stem cell therapy represents a transformative approach in the realm of eye damage repair, particularly for conditions that were once considered irreparable. By harnessing the regenerative capabilities of stem cells, therapies like the CALEC procedure offer a fresh perspective on treating serious ocular surface disorders. The results from recent clinical trials underscore the potential of this cutting-edge treatment, achieving significant restoration of the cornea’s integrity and functionality. As the safety and efficacy of stem cell applications in ophthalmology become increasingly validated, we anticipate a shift in how clinicians manage severe eye injuries.
Furthermore, stem cell clinical trials pave the way for expanding therapeutic options for patients with diverse ocular pathologies. Continuous advancements in manufacturing stem cell grafts and refining transplant procedures will undoubtedly improve patient outcomes and broaden the scope of treatable conditions. This novel approach may soon transition from experimental to standard practice, revolutionizing management strategies for eye diseases and enhancing the quality of life for countless individuals affected by eye damage.
The Role of Research Collaborations in Advancing CALEC
Research collaborations have played a pivotal role in the rapid advancement of the CALEC procedure. Through partnerships with esteemed institutions like Dana-Farber and Boston Children’s Hospital, the development and refinement of this innovative treatment became feasible, bridging the gap between laboratory research and clinical application. Collaborative efforts ensure that the CALEC procedure is not only scientifically sound but also meets the rigorous clinical and safety standards required for human use. This synergy among multidisciplinary teams fosters innovation and accelerates the timeline for bringing effective treatments to patients.
The involvement of diverse expertise in the CALEC clinical trial has also contributed to a more thorough understanding of the procedure’s effects and limitations. Feedback and findings gathered along the way are crucial for ongoing research, as they can lead to further improvements and modifications of the treatment protocol. As researchers and clinicians work together, the overarching goal remains clear: to deliver groundbreaking therapies that can significantly enhance the lives of individuals grappling with corneal injuries and chronic vision problems.
Enhancing Patient Selection for Future CALEC Trials
As the CALEC procedure proves successful in initial trials, optimizing patient selection criteria will be vital for future studies. Current protocols necessitate that participants possess at least one healthy eye for stem cell extraction, which limits accessibility for many potential patients. Developing a more flexible approach to patient selection, perhaps by exploring allogeneic options in the future, could significantly broaden the number of individuals who can benefit from this innovative therapy. Adjustments to inclusion criteria will be paramount in addressing the urgent needs of those facing severe corneal damage.
Furthermore, rebuilding the spectrum of patients eligible for CALEC will contribute to a stronger body of evidence regarding the treatment’s long-term effects and outcomes. By involving a more diverse patient population, researchers can gain insights into the therapy’s efficacy across different demographics and varying stages of corneal injury. Such insights pave the way for tailored approaches, ensuring that emerging treatments like CALEC are effective for all individuals suffering from limbal stem cell deficiency.
The Safety Profile of Stem Cell Treatments in Ophthalmology
Safety is a paramount concern in the field of ophthalmology, particularly when considering novel therapies such as stem cell treatments. The CALEC trial has reported a high safety profile, with no severe adverse events documented in the donor or recipient eyes. It is essential for both clinicians and patients to understand the risks versus benefits associated with such groundbreaking procedures. Ongoing monitoring and transparent reporting of safety data ensure that practices remain informed and can adapt to any emerging patterns or complications. As CALEC evolves, the commitment to safety will be crucial as it transitions toward broader clinical application.
In comparison to traditional treatment options, stem cell therapies like CALEC offer a distinctly advantageous safety profile while addressing previously untreatable conditions. The goal is not only to enhance corneal repair but also to minimize the incidence of complications that can arise from conventional surgical interventions. As research continues to validate the outcomes of stem cell therapies in ophthalmology, the provided data will be instrumental in influencing the adoption of these treatments into mainstream practice, ultimately prioritizing patient safety and quality of care.
Future Directions for Stem Cell Research in Eye Care
The expansion of stem cell research in eye care is essential for addressing complex ocular conditions and improving treatment options for patients. As more data emerge regarding the CALEC procedure, future research directions will likely focus on understanding the long-term effects of the treatment as well as refining the procedures involved. Such advancements can include optimizing the cultivation process of stem cells, investigating alternative sources for stem cell extraction, and enhancing transplant techniques. The landscape of ocular therapy is rapidly evolving, and maintaining momentum will require both continued clinical trials and exploration into innovative applications of stem cell technology.
Moreover, interdisciplinary collaborations will be instrumental in pushing the boundaries of what is achievable in eye care. By pooling expertise from various fields, researchers can tackle greater challenges, assess the intricacies of ocular diseases, and develop novel therapies that are both effective and safe. Engaging stakeholders across academia, industry, and regulatory bodies will facilitate an environment conducive to breakthrough discoveries, ultimately translating complex findings into tangible benefits for patients suffering from debilitating eye conditions.
Implications of CALEC on Standard Care Practices
The introduction of the CALEC procedure has significant implications for existing standard care practices in the treatment of corneal injuries. By demonstrating a safe and effective means of restoring corneal surfaces, CALEC challenges the conventional reliance on corneal transplants alone. As clinical trials yield positive results, practitioners may begin to incorporate this innovative therapy into their treatment arsenals, creating new protocols that prioritize personalized stem cell treatments in cases of limbal stem cell deficiency. This evolving standard of care holds the potential to reshape how eye injuries are approached and managed.
Furthermore, the enthusiasm surrounding the CALEC procedure underscores the necessity of ongoing education and training for eye care professionals. As novel treatments develop, practitioners must remain informed about the latest research findings and their implications for clinical practice. This demand for advanced knowledge will not only enhance the quality of patient care but also foster a culture of innovation within the field, paving the way for further breakthroughs that can exponentially improve outcomes for patients suffering from corneal injuries.
The Role of Regulatory Bodies in Advancing Stem Cell Therapies
Regulatory bodies play a key role in overseeing the development and implementation of stem cell therapies in healthcare. For procedures like CALEC, the approval process mandates stringent testing for safety and efficacy to protect patient welfare. Successful navigation of regulatory frameworks is crucial for translating promising experimental treatments into widely accessible therapies. Continuous collaboration between researchers, clinicians, and regulatory entities ensures that advancements in stem cell technology are met with the appropriate guidelines that can facilitate a safe and ethical rollout of new treatment options.
Moreover, regulatory developments can significantly influence public perception and acceptance of stem cell therapies in medicine. As positive trial outcomes are documented and shared, regulatory bodies must also engage in public education regarding the safety and benefits of such treatments. By promoting transparency and fostering trust, these organizations contribute to a more informed patient population ready to embrace innovative therapeutic options designed to enhance health, function, and overall quality of life.
Frequently Asked Questions
What is stem cell therapy for cornea repair?
Stem cell therapy for cornea repair involves using stem cells, specifically cultivated autologous limbal epithelial cells (CALEC), to restore the surface of a damaged cornea. This process includes harvesting stem cells from a healthy eye, expanding them in a lab, and then transplanting them into the affected eye to promote healing and regeneration.
How does the CALEC procedure work in treating corneal injuries?
The CALEC procedure works by extracting limbal epithelial stem cells from a healthy eye, expanding these cells to create a graft, and surgically transplanting this graft into the eye with corneal damage. This procedure aims to restore the cornea’s surface, which is crucial for proper vision.
What types of corneal injuries can stem cell therapy treat?
Stem cell therapy for cornea repair can treat various corneal injuries, including those resulting from chemical burns, infections, and other types of traumatic damage that lead to limbal stem cell deficiency. This is vital for patients who have not responded to conventional corneal transplants.
What are the success rates of stem cell therapy for cornea repair?
Initial clinical trials of stem cell therapy for cornea repair using CALEC have shown a success rate of over 90% in restoring the corneal surface. Meta-analyses reveal that up to 79% of participants achieved complete restoration of the cornea at twelve months.
Is the CALEC procedure safe for patients?
Yes, the CALEC procedure has demonstrated a high safety profile in clinical trials, with no serious adverse events reported. Most complications have been minor and easily resolved, indicating that stem cell therapy can be a safe option for treating corneal injuries.
What qualifications do patients need to undergo stem cell therapy for cornea repair?
To qualify for stem cell therapy for cornea repair, patients typically need to have only one damaged eye from which stem cells can be harvested. This is crucial for the CALEC procedure, as a biopsy of the healthy eye is required to obtain the necessary stem cells.
How long does it take to see results from the CALEC procedure?
Patients undergoing the CALEC procedure for stem cell therapy usually begin to see results within three months, with some achieving complete restoration of corneal function. Follow-up assessments at twelve and eighteen months can indicate even higher rates of success and improvement.
What is the future of stem cell therapy for corneal injuries?
The future of stem cell therapy for corneal injuries looks promising, with plans for larger clinical trials and potential allogeneic approaches, which would allow for stem cells to be sourced from donors. Such advancements could greatly increase access to effective corneal repair treatments.
Are there any ongoing clinical trials for stem cell therapy for cornea repair?
Yes, ongoing clinical trials are being conducted to further assess the efficacy and safety of stem cell therapy for cornea repair. These studies aim to include larger populations and longer follow-up periods, paving the way for possible FDA approval in the future.
Where can I find more information about stem cell therapy for cornea repair?
For more information about stem cell therapy for cornea repair, including ongoing clinical trials and the CALEC procedure, you can visit the Mass Eye and Ear website or consult with specialists in ophthalmology and cornea services.
| Key Point | Details |
|---|---|
| Clinical Trial | Aimed to test stem cell therapy (CALEC) for cornea repair, involving 14 patients followed for 18 months. |
| Procedure Overview | Involves harvesting limbal stem cells from a healthy eye, expanding them, and transplanting into the damaged eye. |
| Effectiveness | 90% effectiveness in restoring corneal surface; 50% complete restoration at three months, increasing to 93% by twelve months. |
| Safety Profile | High safety profile with no serious events; one bacterial infection occurred, resolved quickly. |
| Future Studies | Next trials aim to include larger populations, longer follow-ups, and randomized-control designs. |
Summary
Stem cell therapy for cornea repair is emerging as a groundbreaking solution for patients with corneal damage previously deemed untreatable. The clinical trial conducted at Mass Eye and Ear demonstrated that this innovative approach, utilizing cultivated autologous limbal epithelial cells (CALEC), can effectively restore corneal surfaces with a remarkable success rate of over 90%. This therapy not only addresses visual impairment but also significantly improves the quality of life for affected individuals. As further studies are needed to enhance the scope of this treatment, the future looks promising in making stem cell therapy widely accessible, heralding a new era in ophthalmic care.